Suberoyl chloride
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name Octanedioyl dichloride | |
| Other names Suberoyl dichloride; Suberic acid chloride | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.156.463 |
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
InChI
| |
| |
| Properties | |
Chemical formula | C8H12Cl2O2 |
| Molar mass | 211.08 g·mol−1 |
| Density | 1.172 g/cm3 |
| Boiling point | 162–163 °C (324–325 °F; 435–436 K) |
Solubility in water | Reacts with water |
| Hazards | |
| GHS labelling: | |
Pictograms |  |
| Danger | |
Hazard statements | H314 |
Precautionary statements | P260, P264, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P363, P405, P501 |
| Flash point | 110 °C (230 °F; 383 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). Infobox references | |
Chemical compound
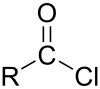
Suberoyl chloride is an organic compound with the formula (CH2)6(COCl)2. It is the diacid chloride derivative of suberic acid. It is a colorless liquid although aged samples appear yellow or even brown.
Uses
Suberoyl chloride is used as a reagent to synthesize hydroxyferrocifen hybrid compounds that have antiproliferative activity against triple negative breast cancer cells. It is also used as a cross-linking agent to cross-link chitosan membranes, and also improves the membrane's integrity.[1]
References
- ^ "Suberoyl chloride". Alfa Aesar. Retrieved 16 April 2019.
External links
- MSDS Safety data (PDF)
- v
- t
- e
Diacyl chlorides (-COCl)2
- Oxalyl chloride C2O2Cl2
- Malonyl chloride C3H2Cl2O2
- Succinyl chloride C4H4Cl2O2
- Glutaryl chloride C5H6Cl2O2
- Adipoyl chloride C6H8Cl2O2
- Pimeloyl chloride C7H10Cl2O2
- Suberoyl chloride C8H12Cl2O2
- Azelaoyl chloride C9H14Cl2O2
- Sebacoyl chloride C10H16Cl2O2
Category:Acyl chlorides
 | This article about an organic compound is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e












