Cobalt germanide
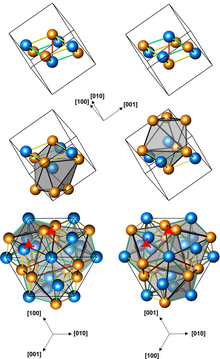 Structures of left-handed and right-handed cubic CoGe crystals (3 presentations, with different numbers of atoms per unit cell; orange atoms are Ge) | |
| Names | |
|---|---|
| IUPAC name Cobalt germanide | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
PubChem CID |
|
CompTox Dashboard (EPA) |
|
InChI
| |
| |
| Properties | |
Chemical formula | CoGe |
| Molar mass | 131.56 g/mol |
Magnetic susceptibility (χ) | 1.3×10−6 emu/g[1] |
| Structure | |
Crystal structure | Monoclinic[1] |
Space group | C2/m (No. 12), mS16 |
Lattice constant | a = 1.165 nm, b = 0.3807 nm, c = 0.4945 nm α = 90°, β = 101.1°, γ = 90° |
Formula units (Z) | 8 |
| Hazards | |
| Flash point | Non-flammable |
| Related compounds | |
Other anions | Cobalt silicide |
Other cations | Iron germanide Manganese germanide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).  N (what is N (what is  Y Y N ?) N ?) Infobox references | |
Chemical compound
Cobalt germanide (CoGe) is an intermetallic compound, a germanide of cobalt.
Cubic CoGe crystals (space group P213, cP8, a = 0.4631 nm) can be produced by processing a mixture of Co and Ge powders at a pressure of 4 GPa and a temperature of 800–1000 °C for 1 to 3 hours. They have no inversion center, and are therefore helical, with right-hand and left-handed chiralities. The cubic CoGe is metastable, and converts into a monoclinic phase upon subsequent heating to 600 °C at ambient pressure.[1]
Cubic CoGe is an antiferromagnet with a transition temperature Tc of 132 K.[1]
References
- ^ a b c d Takizawa, H.; Sato, T.; Endo, T.; Shimada, M. (1988). "High-pressure synthesis and electrical and magnetic properties of MnGe and CoGe with the cubic B20 structure". Journal of Solid State Chemistry. 73 (1): 40–46. Bibcode:1988JSSCh..73...40T. doi:10.1016/0022-4596(88)90051-5.
- v
- t
- e
Cobalt compounds
- HCo(CO)4
- CoBr2
- Co(CN)2
- CoCO3
- CoC2O4
- CoCl2
- Co(ClO3)2
- Co(ClO4)2
- CoF2
- Co(HCO2)2
- CoI2
- Co(NO3)2
- Co3(PO4)2
- Co(OAc)2
- CoGeO3
- CoO
- Co(OH)2
- CoS
- Co(OCN)2
- Co(SCN)2
- CoSO4
- CoSe
- Co3P2
- CoH2
- Co(C3H6O3)2
- C
24H
48CoO
4 - C
36H
70CoO
4
- CoSi
- CoGe
- Co3O4
- CoAs
- CoCl3
- Co(NO3)3
- Co2O3
- CoF3
- Co(OH)3
- LiCoO2
- NaxCoO2
- CoF4
- Cs2CoF6
- CoC28H44
- Na3CoO4












 Cobalt_germanide
Cobalt_germanide