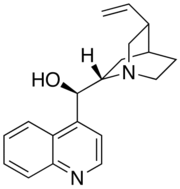
Cinchonidine
 | |
| Names | |
|---|---|
| IUPAC name (9R)-8α-Cinchonan-9-ol | |
| Systematic IUPAC name (R)-[(2S,4S,5R)-5-Ethenyl-1-azabicyclo[2.2.2]octan-2-yl](quinolin-4-yl)methanol | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
Beilstein Reference | 89690 |
| ChEBI |
|
| ChEMBL |
|
| ChemSpider |
|
| ECHA InfoCard | 100.006.930 |
| EC Number |
|
| KEGG |
|
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
InChI
| |
| |
| Properties | |
Chemical formula | C19H22N2O |
| Molar mass | 294.43 g/mol |
| Density | 1.2 g/mL |
| Melting point | 204 to 205 °C (399 to 401 °F; 477 to 478 K) |
| Boiling point | 464.5 °C (868.1 °F; 737.6 K) |
Solubility in water | slightly soluble 0,25 g·l−1 (20 °C) [1] |
| Hazards | |
| GHS labelling: | |
  | |
| Warning | |
| H302, H317, H361, H373 | |
| P201, P202, P260, P261, P264, P270, P272, P280, P281, P301+P312, P302+P352, P308+P313, P314, P321, P330, P333+P313, P363, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).  Y verify (what is Y verify (what is  Y Y N ?) N ?) Infobox references | |
Chemical compound
Cinchonidine is an alkaloid found in Cinchona officinalis and Gongronema latifolium.[2] It is used in asymmetric synthesis in organic chemistry.
References
- v
- t
- e











